Every June and July Ukraine starts harvesting winter wheat. The war with Russia may complicate things this year.
You see, Ukraine is one of the world’s largest agricultural producers and exporters. The war is preventing Ukrainian farmers from harvesting and storing their grain. And closed ports are preventing them from shipping it to countries desperate for food. The ripple effects are already being felt across the globe.
How bad will it get? Only time will tell.
The war in Ukraine is forcing world leaders and scientists to reevaluate how we are meeting the needs of the world’s expanding population, which the UN expects to reach 9.8 billion by 2050.
CRISPR,1CRISPR stands for Clustered Regularly Interspaced Short Palindromic Repeats. a relatively new gene editing technology, could be the key to unlocking tremendous potential in food production.
Agriculture as Innovation
Throughout history, the solution to hunger has often come at the hand of innovation. In 1968, Norman Borlaug, a US scientist, developed a strain of wheat that increased crop yields. In the 70s, Yuan Longping, a Chinese scientist, developed a strain of rice that helped “feed the world.” His rice increased rice yields by 20 percent.
These scientists’ innovations have fed billions of people and directly reduced global poverty.
Agriculture is innovation. From the creation of fields and plows, to Borlaug and Longping, to the invention of GMOs, agricultural innovations have kept pace with the world’s growing population.
And now CRISPR. At only 10 years old, this gene editing technology has endless potential to innovate agriculture, further extending our ability to feed the growing population as well as mitigate the risks to food supply caused by climate change.
In 2020 Jennifer Doudna and Emmanuelle Charpentier received a Nobel Prize in Chemistry for their work on CRISPR. In her keynote speech on World CRISPR Day, Doudna cited agriculture and climate change as the most promising ways CRISPR can help humanity.
This article will examine the potential benefits of CRISPR and how we should consider risk when thinking about regulation. Gene editing’s enormous potential for good should be weighed against its risks. Regulators should mitigate the most serious risks, but otherwise stay out of the way so that innovation can flourish.
What is CRISPR?
CRISPR technology works like a set of scissors for DNA. It searches for a DNA sequence and then clips it, shutting off that specific sequence. First characterized in 1993, CRISPR was developed into a gene editing tool in 2012 by Doudna and Charpentier.
Since its discovery, CRISPR has been revolutionary. Some practical agricultural applications already underway include mushrooms that don’t brown, more nutrient-dense tomatoes, oranges resistant to “citrus greening” disease, cacao plants resistant to disease and climate change, higher-yielding corn and rice, and so much more.
GMOs vs Gene Editing
GMOs (genetically modified organisms) are created when the DNA of an organism is changed by the introduction of DNA from a different species or organism. Concern over GMOs has led to laws about labeling and institutions dedicated to creating a non-GMO food supply. Due to public scrutiny, GMOs have been over-regulated and are not used as much as they could be.
Critics worry that GMOs will have negative health consequences. They also worry about increases in pesticides in GMO foods. There is little evidence that any of this is true. GMOs have actually reduced the need for pesticides.
While these are legitimate concerns, the worst outcomes have not materialized, and the use of GMOs has allowed us to feed the growing population and prevent disasters in food production.
For example, in the 90s, the Hawaiian papaya was being decimated by a virus, reducing its production by 40 percent. In 1997, scientists modified the papaya to be resistant to the virus. Since then, production has completely returned.
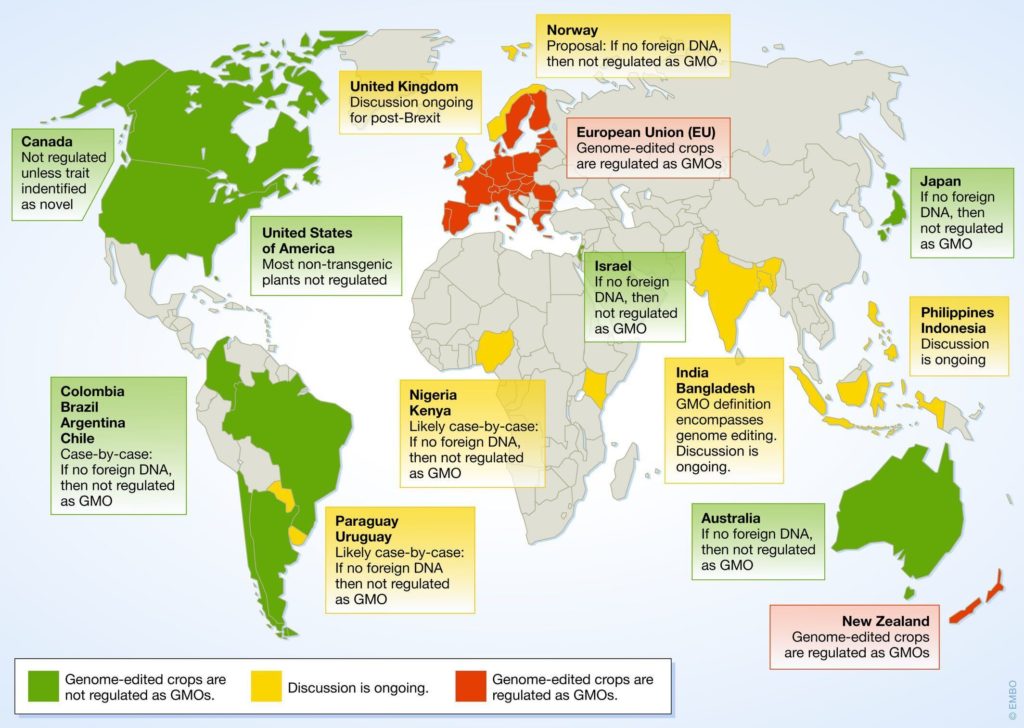
Inside the US, GMO regulation has largely remained unrestrictive. This is not the case in the European Union. Inside the EU there is only one GMO that has been approved for cultivation, and the license has since expired and is waiting for reapproval.
Although similar, GMOs and CRISPR are different processes. CRISPR and other forms of gene editing involve editing existing DNA without inserting foreign DNA. Through gene editing, scientists can select certain DNA to be removed. This process is one that happens over time in nature and is similar to selective breeding which can produce more desired offspring.
Today many foods created through selective breeding are available for public consumption, including chickens that produce larger eggs. CRISPR is similar to selective breeding. We don’t need to fear it.
Promise and Peril of Gene Editing
The first CRISPR edited food on the market was a tomato. In December 2021, tomatoes in Japan that had been edited to contain higher amounts of GABA, a nutrient that is believed to promote lower blood sugar and increase relaxation, were first sold for consumption.
A study in China found that using CRISPR to edit certain genes in rice could increase yields 25–31 percent. Environmentally and economically, this means more rice per acre of land, helping us make better use of the earth around us.
CRISPR can be used in many different industries, not just agriculture. Here are a few companies trying to solve big problems using CRISPR:
- CRISPR Therapeutics is “focused on developing transformative gene-based medicines for serious human diseases.”
- Plantedit is trying to increase the world’s food supply using CRISPR.
- Prime Medicine raised $315M in 2021 to use CRISPR to cure rare diseases.
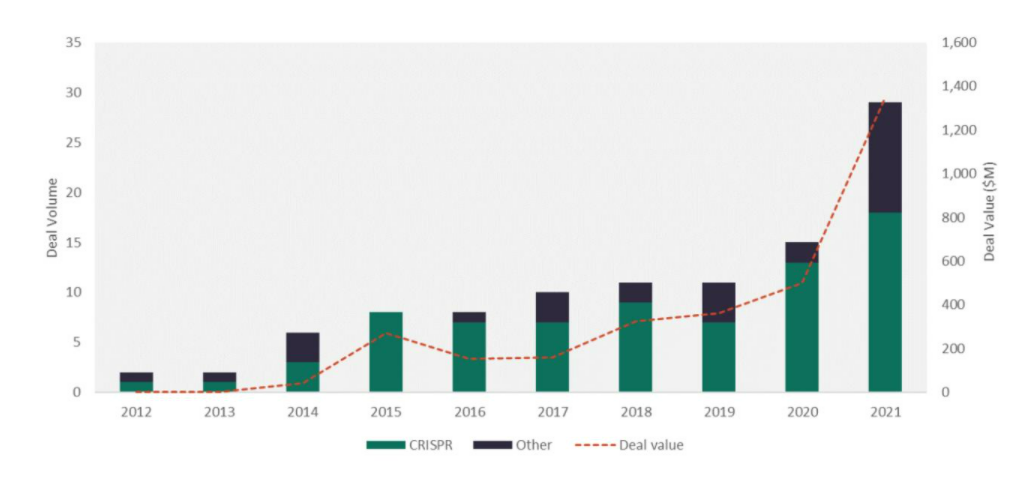
In 2021 alone, over $1.2B was poured into gene editing companies via private investments, an increase of more than 250% from 2020.
Figure 1: The Number and value of gene editing VC deals has increased since 2011
Source: GlobalData Pharmaceutical Intelligence Center (Accessed 20 January 2022)
Scientists at the University of California, Davis, have used CRISPR to edit the embryo of a bull calf named Cosmo to produce more male offspring. Male cattle are 15 percent more efficient at converting food into weight gain. This could help the environment as fewer cattle needed for the same amount of consumption means less space for grazing, less water and food, and less methane released into the atmosphere.
While the possibilities of CRISPR remain endless, downsides still exist.
One of the risks associated with CRISPR is the potential for unintended consequences of editing a specific gene. CRISPR could theoretically cause an irreversible change in the species that has been edited. In the gene editing space, there are two types of editing: germline and somatic.
Germline editing is performed on eggs, sperm, and embryos and can be passed down to future generations. This has the potential to radically improve the human condition by eliminating certain inheritable diseases but could be dangerous otherwise.
Somatic editing only alters the individual species being treated, and therefore won’t be passed on to future generations.
Today most of the genome editing going on is done somatically, which is less risky. Germline has the potential to cause irreversible changes to DNA, which could have unintended consequences on the host species. Somatic editing gives us more room for error.
Current Regulation in the US and EU
In the EU there are stringent regulations required for crops before they can be commercialized. This needs to be changed, as scientists have noted in Genome Biology,
“The current legal framework within the EU regulates CRISPR crops as GMO crops, even when the products are transgene-free and contain no foreign DNA. These regulatory frameworks increase the time and cost of variety development. Scientists and policymakers need to work together and devise comprehensive plans for CRISPR crops integration.”
Research has found no evidence that the use of GMOs, especially in our food, causes damage or danger to humans. Although gene editing technologies differ from GMOs, inside the EU they are currently regulated the same as GMOs. They should not be treated the same, and this regulation needs to change to allow for innovation.
Source: “The Evolving Landscape around Genome Editing in Agriculture,” EMBO Reports 21, no. 6 (June 4, 2020): e50680, https://doi.org/10.15252/embr.202050680.
The US has struck a good balance in the regulation of gene editing used for agricultural products. Currently the US regulates gene editing as different from GMOs. The secretary of agriculture issued the following statement in 2018:
“[The] USDA has no plans to regulate plants that could otherwise have been developed through traditional breeding techniques as long as they are not plant pests or developed using plant pests.”
The deregulation of this sector has led to many positive impacts on our food supply and will continue to do so.
While the US is keeping the regulatory burden low for crops, it is strict regarding humans and animals.
In a recent survey about getting CRISPR products to clinical trials, 47 percent of researchers said their biggest issue was regulatory. This is a problem, large organizations that regulate industries usually lag the development of new technology. For example, to get FDA approval for a new drug takes on average 12 years.
There are benefits to making sure the new technology is safe, but to what extent does unnecessary regulation halt innovation? Unnecessary regulatory burdens can cause less innovation and put a damper on the possible success of these technologies.
Conclusion
If CRISPR is allowed to grow, it has the potential to save lives, better our environment, and feed the world. For this to happen, regulatory institutions need to regulate only the most serious risks with the goal of allowing the technology to move forward.
The greatest peril of new gene editing in agriculture is the risk that we fail to take advantage of it, leaving millions exposed to unnecessary food insecurity.